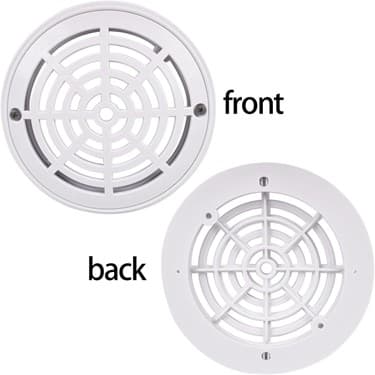
TopHomer Pool Drain Covers Recalled Over Drowning Hazards
TopHomer recalled pool drain covers on December 18, 2025, due to serious entrapment and drowning hazards. The recall affects 8-inch drain covers sold on Amazon. Consumers should immediately stop using the products and seek refunds.