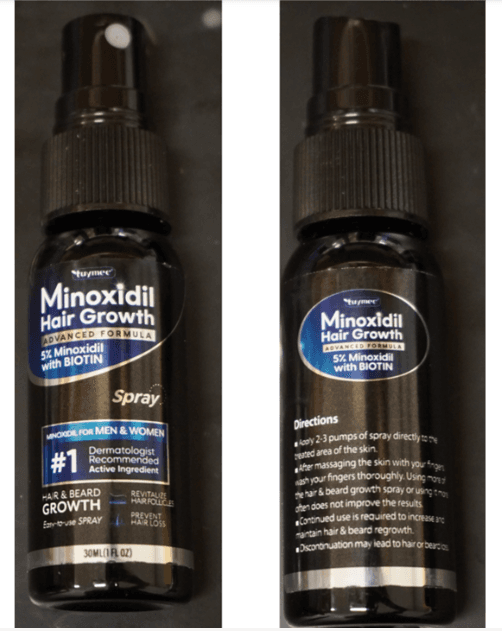
Vitaquest International Recalls Multiple Iron Supplements for Child-Resistant Packaging Violations (
Vitaquest International recalled Arey Not Today iron supplements sold at retailers nationwide after determining the products do not meet the Poison Prevention Packaging Act's child-resistant packaging requirement. The noncompliant packaging creates a poisoning risk to young children if ingested. Consumers should stop using the products immediately and contact Vitaquest for a free child-resistant替换